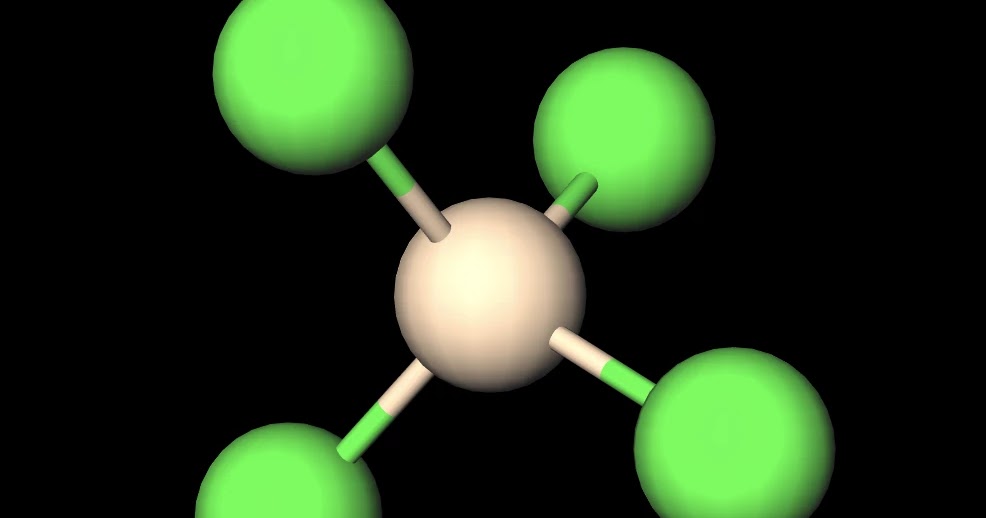
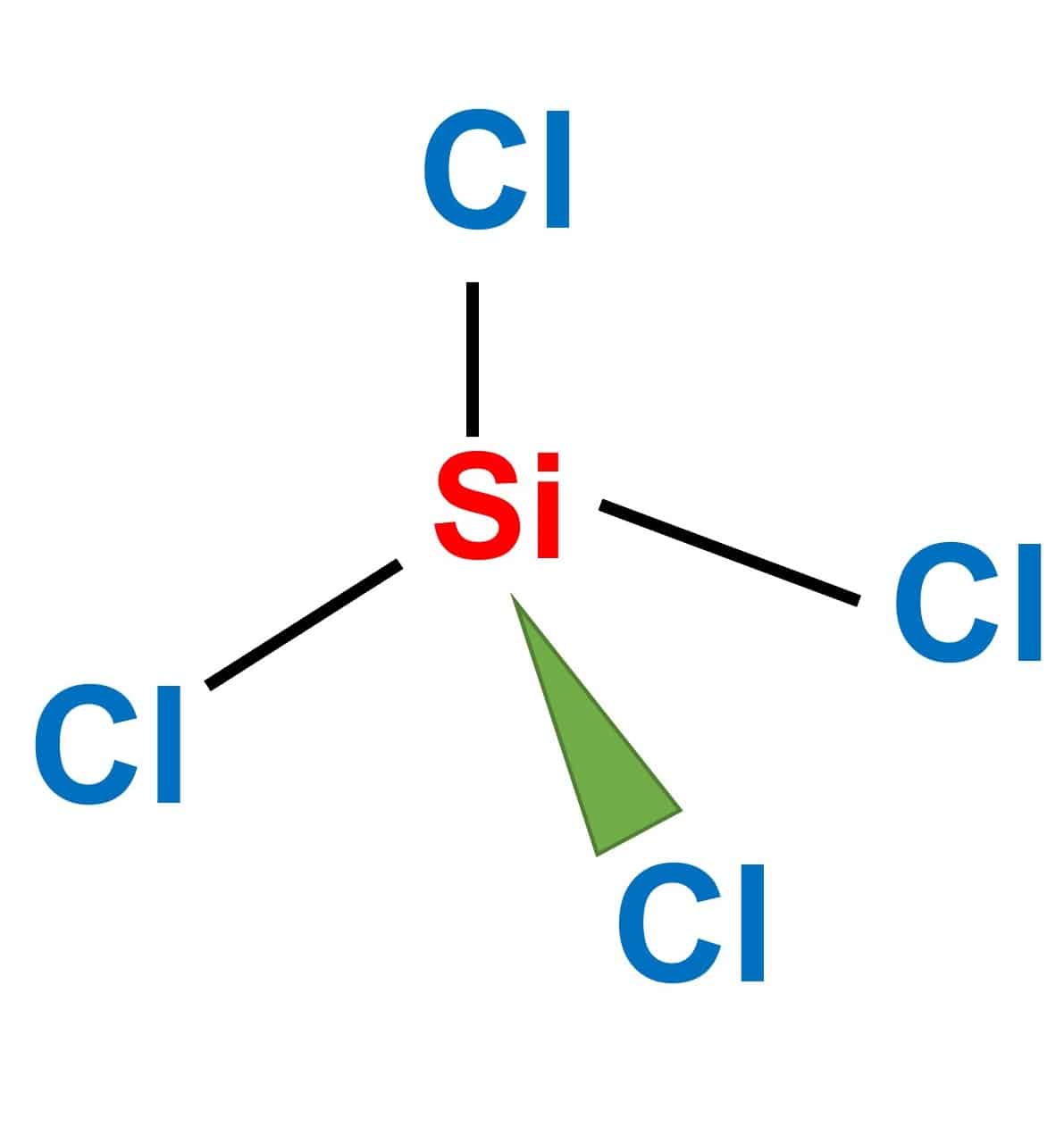
Draw the Lewis structure for SiCl4. How many bonds are around the central atom, and what is the shape of this molecule? | Homework.Study.com

Is SiCl4 Polar or Non-polar? (Silicon Tetrachloride) | Is SiCl4 Polar or Non-polar? (Silicon Tetrachloride) We know the concept of polarity is a bit tricky and one might need a little practice

NHC→SiCl4: An Ambivalent Carbene‐Transfer Reagent - Böttcher - 2015 - Chemistry – A European Journal - Wiley Online Library

The SiCl4 molecule is nonpolar and chlorine is more electronegative than silicon. From this information alone it can be deduced that :(1) Si - Cl bond is nonpolar(2) SiCl4 molecule is planar(3)

All four of the silicon-chlorine single bonds in sicl4 are polar. in which direction should the polarity - brainly.com

Assertion: A molecule of silicon tetrachloride (SiCl4) is nonpolar.Reason: The four bonds in SiCl4 are identical and the molecule has a tetrahedral structure.
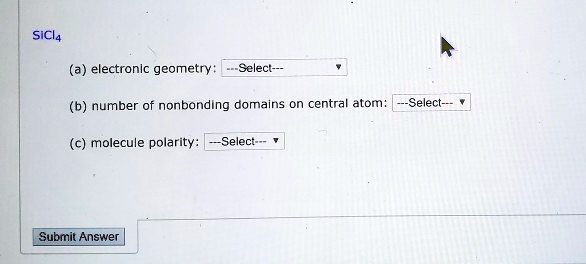
SOLVED: SiCl4 Electronic Geometry: Select (b) Number of Nonbonding Domains on Central Atom: Select Molecule Polarity: Select

SiCl4 lewis structure, molecular geometry, hybridization, polar or nonpolar | Molecular geometry, Molecular shapes, Molecular