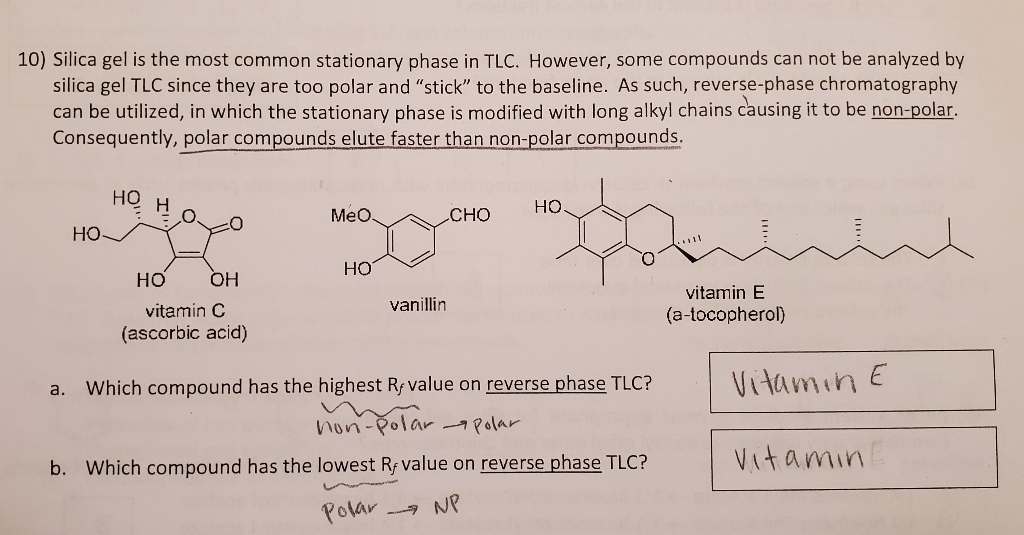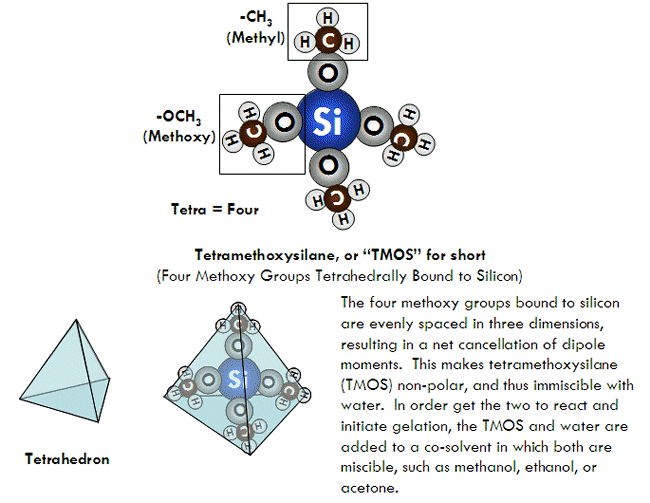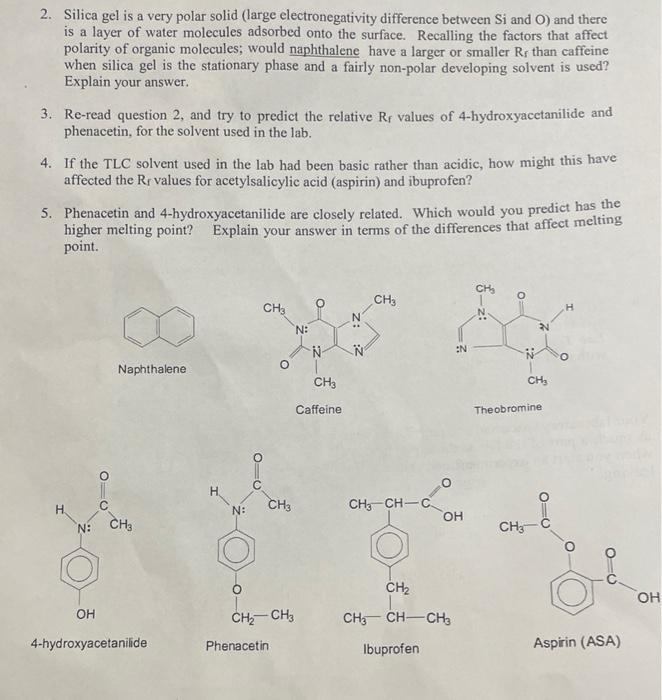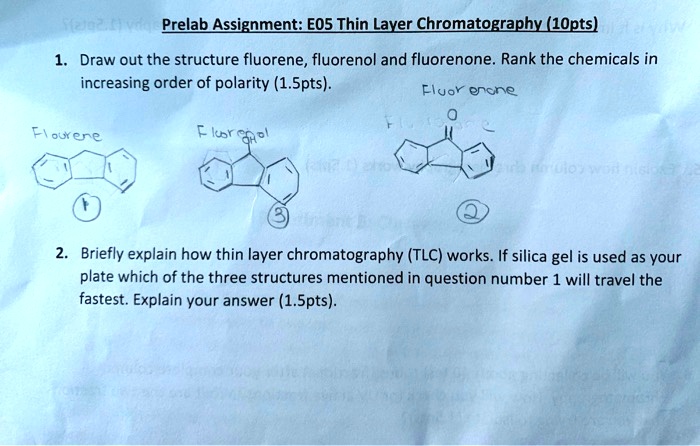
SOLVED: Prelab Assignment: EOS Thin Layer Chromatography (10 pts) Draw out the structure of fluorene, fluorenol, and fluorenone. Rank the chemicals in increasing order of polarity (10 pts). Briefly explain how thin
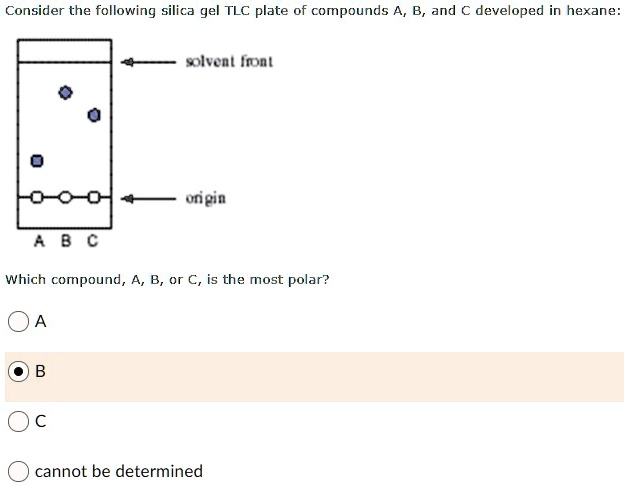
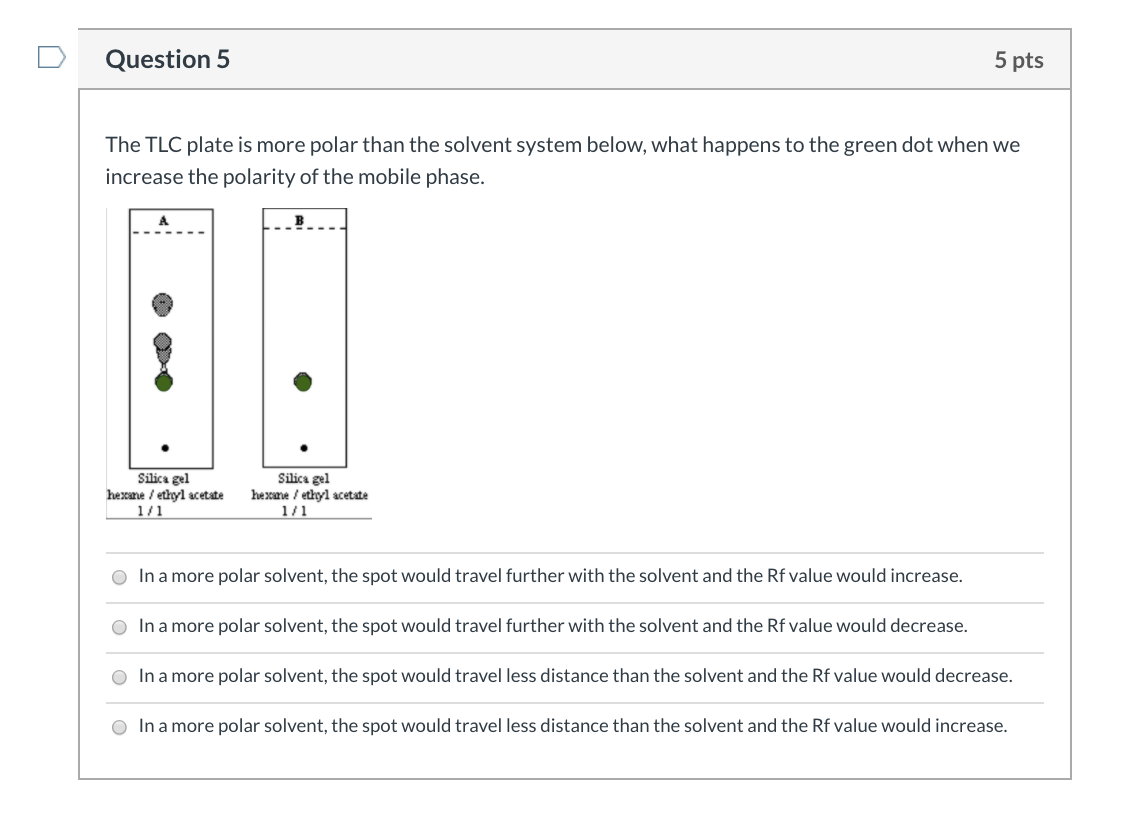
SOLVED: Consider the following silica gel TLC plate of compounds A and C developed in hexane: solvent Front Rf value Which compound, A or C, is the most polar? cannot be determined

A Mild Silica Gel Promoted Synthesis and Initial Functional Study of Tetrapyridyl Tetrahydropyrrolopyrrolones | Organic Letters
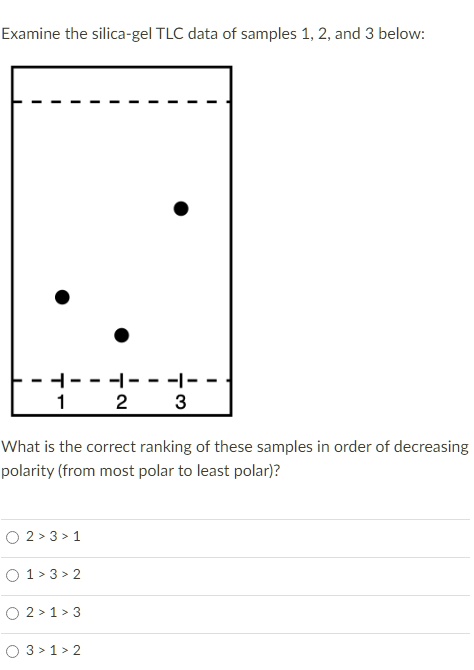
SOLVED: Examine the silica-gel TLC data of samples 1, 2, and 3 below. What is the correct ranking of these samples in order of decreasing polarity (from most polar to least polar)?
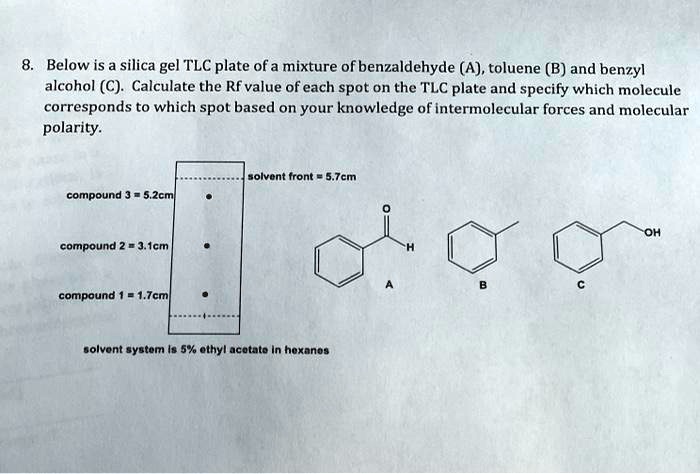
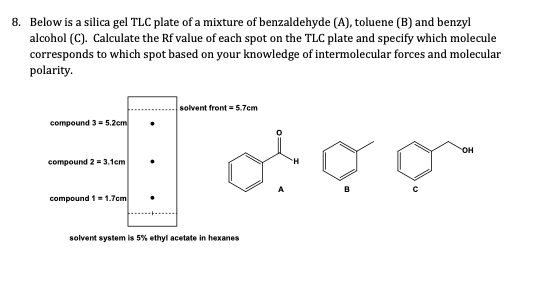
SOLVED: Below is a silica gel TLC plate of a mixture of benzaldehyde (A), toluene (B), and benzyl alcohol (C). Calculate the Rf value of each spot on the TLC plate and

The impact of the silica gel structure and surface chemistry on the melting of aliphatic nanocrystals: Thermodynamic model and experiment - ScienceDirect

.png?revision=1&size=bestfit&width=1103&height=424)