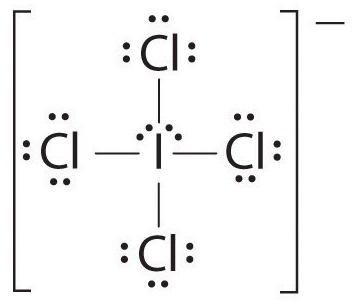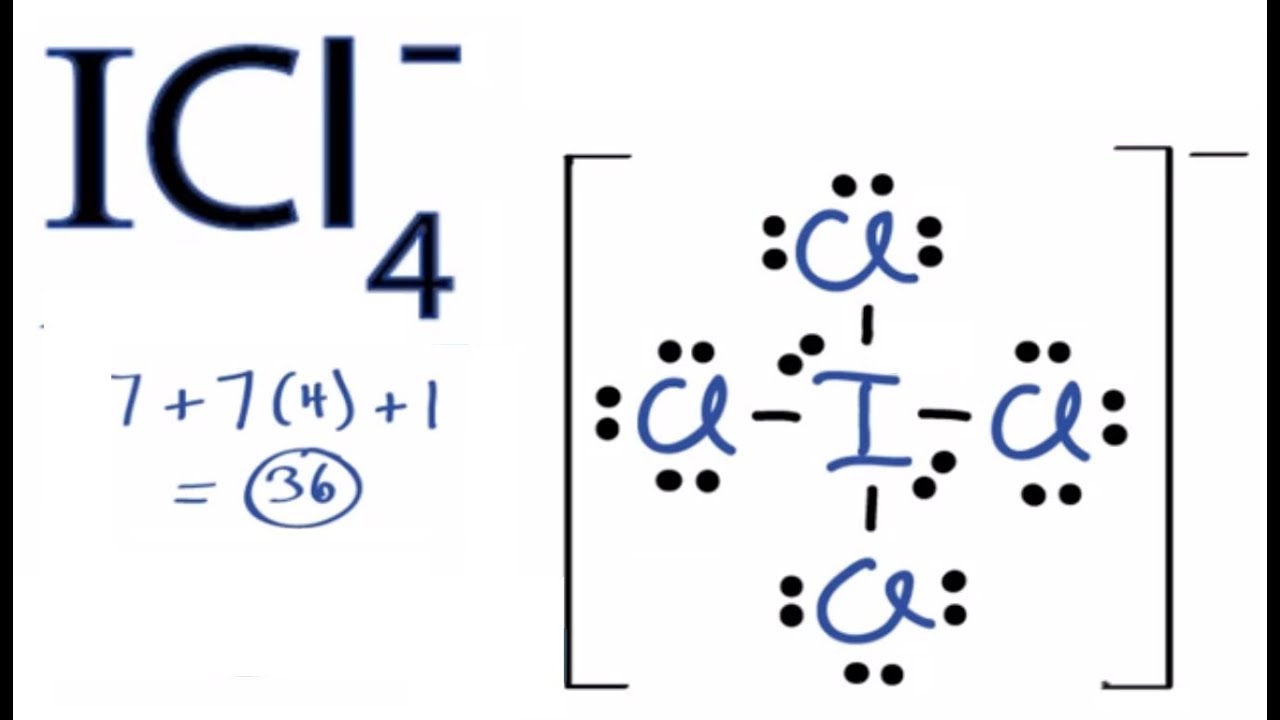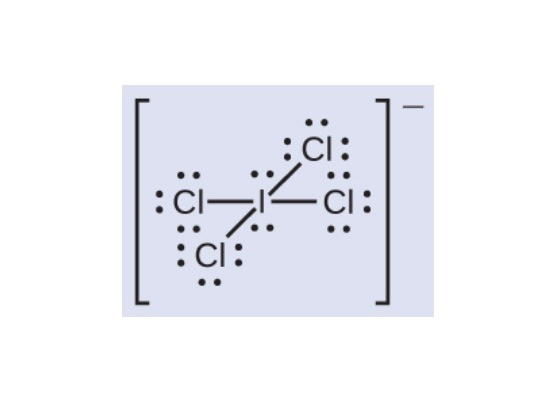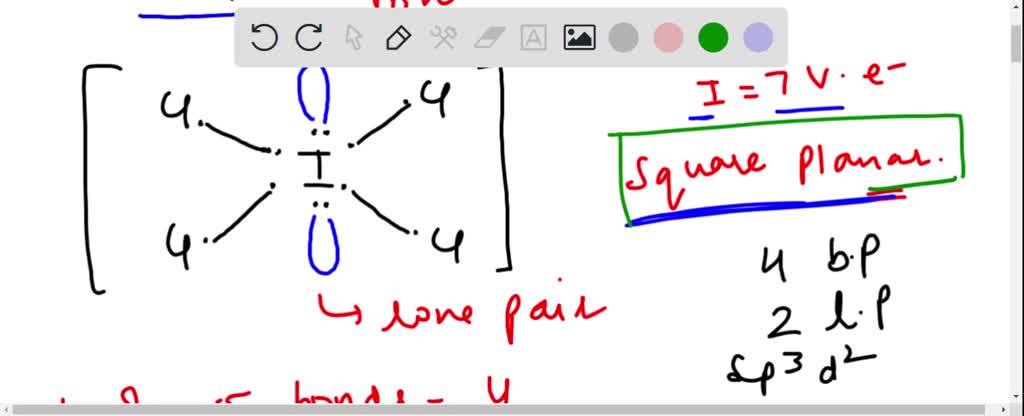
SOLVED: ICl4- Polar or Nonpolar VSEPR name: a. octahedral b. tetrahedral c. trigonal bipyramidal d. square planar Hybridization: a. sp2 b. sp3d2 c. sp3d d. sp3

SOLVED: Molecular Structure: The VSEPR Model Examples: Moment of Eq = Predict shape and polarity A. polar (μ ≠0) B. nonpolar (μ = 0) PCl3 Vs BCl3 PCl5 Vs SbCl5 SF4

SOLVED: The number of lp - bp repulsion present in ClF3 at nearly 90 degree angle are : A. 1 B. 2 C. 3 D. 4