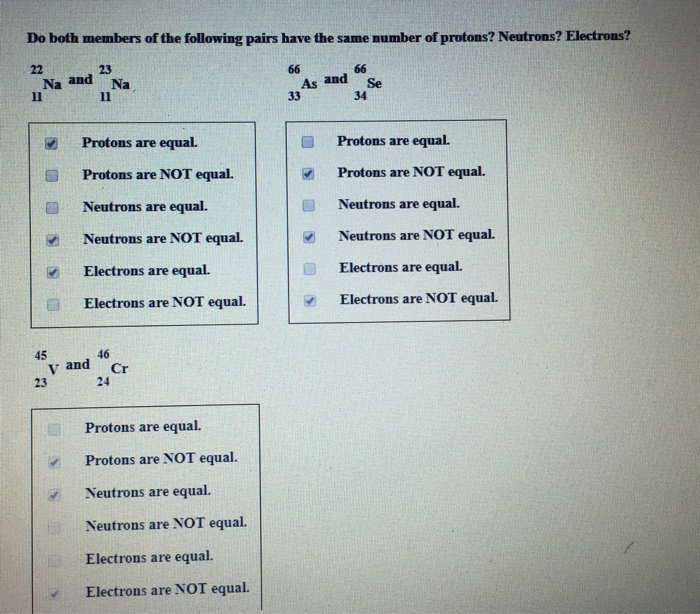
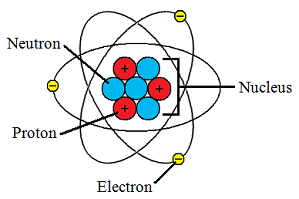
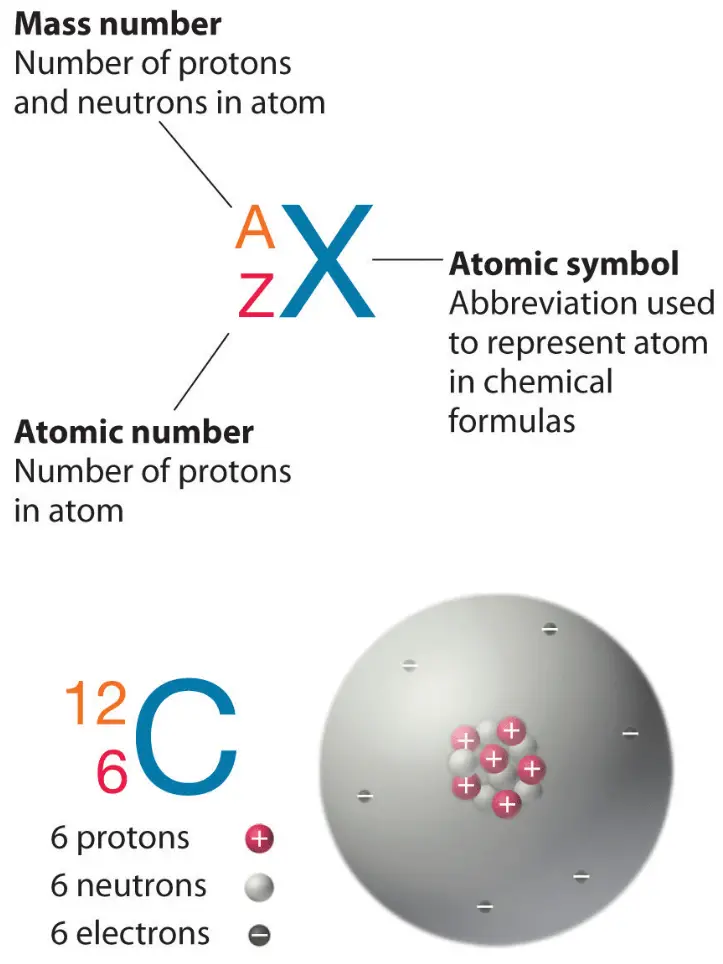
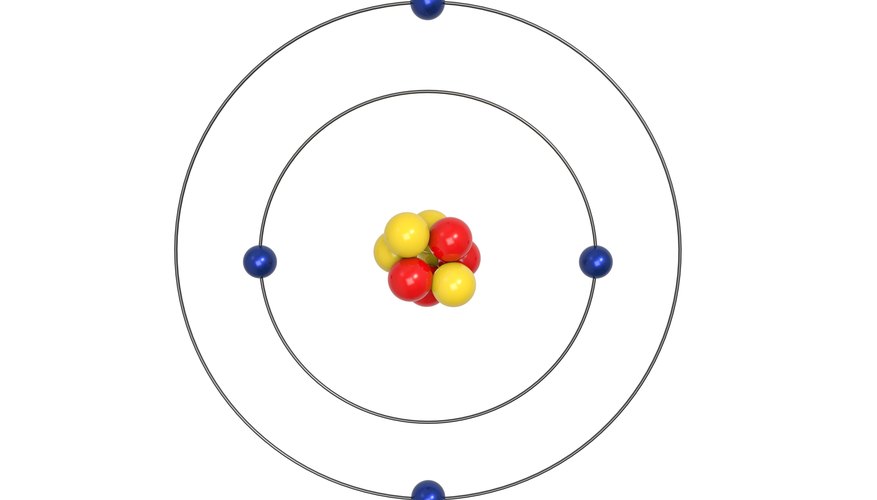
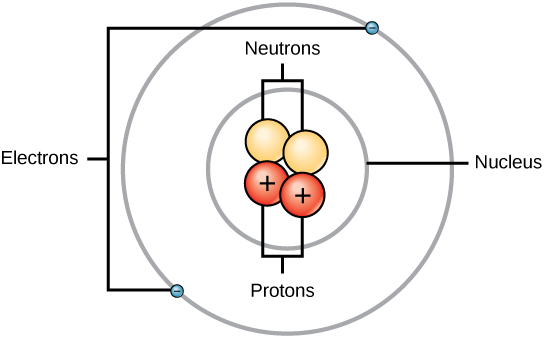
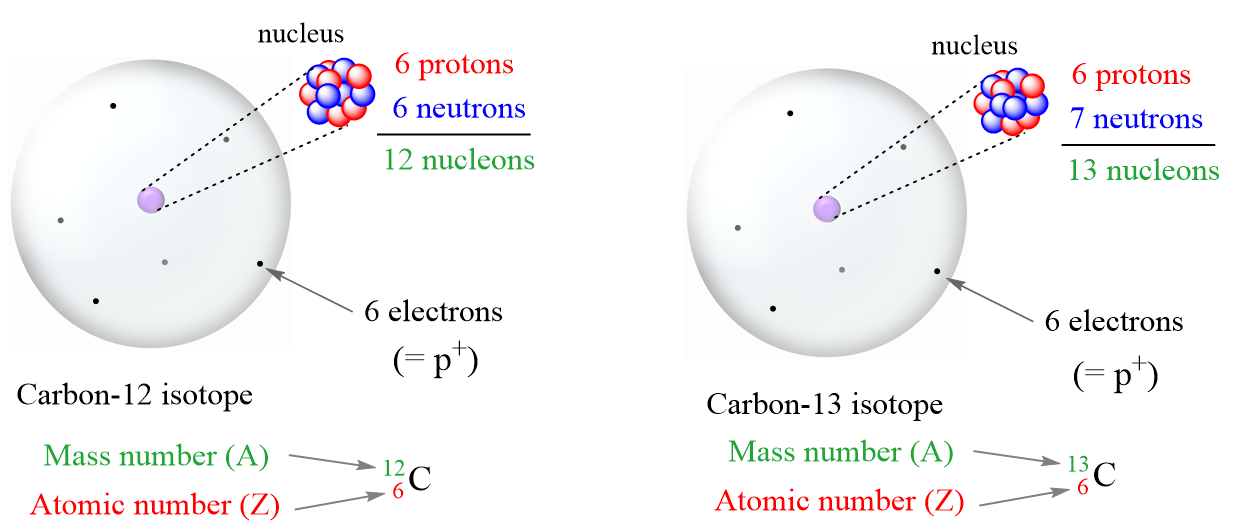
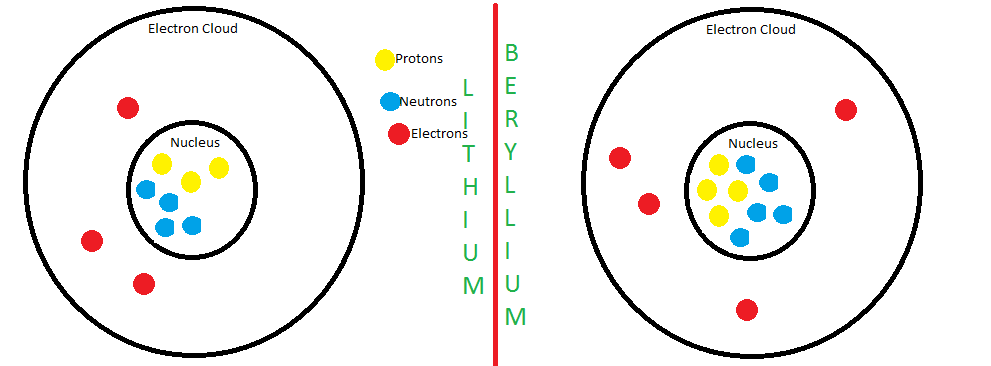
The atomic number equals the number of protons. The electrons in a neutral atom equal the number of protons. The mass number equals the sum of. - ppt download

Question Video: Reading Number of Electrons, Protons, and Neutrons from Atomic Number and Mass Number | Nagwa
If the count of no. of protons is equal to Atomic number, no. of protons, no. of neutrons, no.of electrons in Sodium, Magnesium are?